WHAT ARE THE ELEMENTS IN THE CIRCLED AREA CALLED?


WHICH ATOM HAS THE LARGER IONIZATION ENERGY?
Mg (Magnesium) or N (Nitrogen)


WHAT'S THE SYMBOL FOR AN ION THAT CONTAINS 34 PROTONS AND 36 ELECTRONS?

WHAT IS THE TOTAL NUMBER OF VALENCE ELECTRONS IN AN ATOM OF Ba?

WHAT IS THE DEFINITION OF ELECTRON AFFINITY?

COMPLETE THE SENTENCE:
REMOVING THE SPECTATOR IONS OF AN EQUATION LEAVES YOU WITH
THE ?

WHAT IS THE OXIDATION NUMBER OF Mg IN THE COMPOUND MgO?

HOW MANY ELECTRONS MUST BE ADDED, AND TO WHAT SIDE DO YOU ADD ELECTRONS TO BALANCE AND COMPLETE THE HALF-REACTION?
Cl2 → 2 Cl-

IN THE FOLLOWING HALF-REACTION, WHICH REACTANT IS BEING REDUCED?
Mg(s) + Cl2 (g) → MgCl2 (s)

Would you use an oxidizing agent or reducing agent in order for the following reactions to occur?
Mn2+  MnO2
MnO2

WHAT IS THE NAME OF THE FORMULA?


WHAT IS THE FORMULA OF Tetraphosphorus Hexoxide ?

WHAT'S THE FORMULA OF ANTIMONY PENTAFLOURIDE?

WHAT'S THE NAME OF THE FORMULA?


IS THE FOLLOWING COMPOUND IONIC OR COVALENT ?
NH4OH

TRUE or FALSE?
TEMPERATURE HAS NO EFFECT ON EQUILIBRIUM.

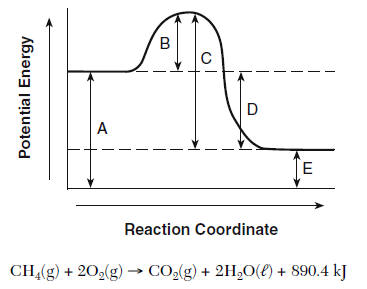
EXOTHERMIC OR ENDOTHERMIC REACTION?


WHAT IS THE SIGN (+/-) ON ΔG FOR A SPONTANEOUS PROCESS?

TRUE OR FALSE?
AT EQUILIBRIUM THE CONCENTRATIONS OF REACTANTS AND PRODUCTS NO LONGER CHANGE.

WHICH LETTER REPRESENTS THE ACTIVIATION ENERGY OF THE REACTION?

WHAT ARE THE NAMES OF THE THREE SUBATOMIC PARTICLES ?

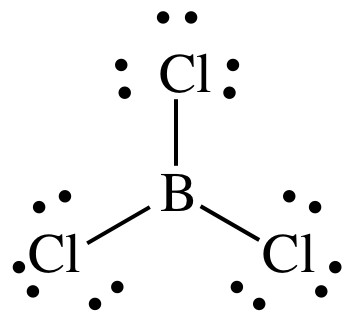
PREDICT THE THREE DIMENSIONAL SHAPE OF THE FOLLOWING MOLECULE.

What is 32°C in Kelvin?
(3 Sig Figs)

PHYSICAL or CHEMICAL CHANGE?
DECOMPOSING WATER BY PASSING AN ELECTRIC CURRENT THROUGH IT.

WHAT DOES THE Z REPRESENT IN THE ISOTOPE NOTATION?


THE NEUTRAL ATOM'S LIKLIHOOD OF GAINNING AN ELECTRON.
ADD 2 ELECTRONS TO THE REACTANT (RIGHT) SIDE
Cl2 + 2 e- → 2 Cl-
CHLORINE GAS (CL2) IS BEING REDUCED.
NEGATIVE
-ΔG = SPONTANEITY
PROTON, NEUTRON, ELECTRON









