What are the three parts of a hypothesis?

What in class activity served as a model for the unknowns of science (e.g., deep space, atomic structure, origin of life on earth, etc.)?

What is a simple representation that allows scientists to explain their observations and make predictions?

What two branches of science fall under the umbrella of Physical Science?

Who said ESSENTIALLY ALL MODELS ARE WRONG, BUT SOME ARE USEFUL?

What is the center of the atom called?

What is the name of the negatively charged particle inside of an atom?

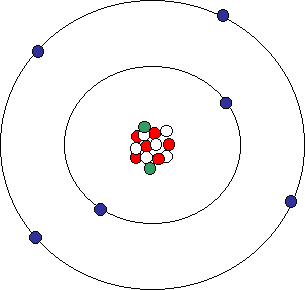
Whose model of the atom is pictured below?

Who had an atomic model that was like PLUM PUDDING or CHOCOLATE CHIP COOKIE DOUGH

What was Murray Gell-Mann's contribution to atomic theory?

Are periods vertical or horizontal?

What is the name of the vertical columns on the periodic table?

What gets added when you move down to the next period?

What is the name of the family with completely filled electron shells?

What makes chlorine a violent element?

What type of mixture is the same throughout?

What is one sign of a chemical change?

Name a type of physical change.

What is name of the phase change for when a solid turns to a gas?

What is the name of the theory that describes the motion of elements in the different states of matter?

Which physical property does not depend on the amount of substance? Intensive or extensive?

What is the formula for density?

What is the name for the temperature when a substance goes from a liquid to a gas?

What is the density of a substance with a mass of 5 grams and a volume of 2 ml?

What is the mass of 6 ml of barium if the density of barium is 3 g/ml?

If... Then... Because...
THE BOX!
Model
Chemistry and Physics
George E. P. Box
Nucleus
Electron
Bohr
J. J. Thomson
Quarks
Horizontal
Groups (or Families)
An Electron Shell
Noble Gases
It needs an electron to have a full shell, so it will take the electron from anywhere. (It'll take an electron from kittens)
Homogeneous
New substance, color change, gas formed, temperature change, light given off
Breaking, bending, mixing, phase change
Sublimation
Kinetic Molecular Theory
Intensive
D = m/v
Boiling point
2.5 ml
18 g










